Introduction
Survival rates for people with mantle cell lymphoma (MCL) have improved in recent years. Among the potential factors accounting for this improvement is the approval of multiple new therapies over the past two decades, including bortezomib, bendamustine, ibrutinib, lenalidomide, acalabrutinib, and zanubrutinib. We hypothesized that the availability of multiple agents with non-overlapping mechanisms of action would result in significant improvements in overall survival following first-line therapy.
Methods
Patients diagnosed with MCL between 2002 and 2017 were identified using the SEER-Medicare linked database. Selected patients were >65 years of age at the time of diagnosis with MCL. Lines of therapy were determined using billing codes, with inpatient/outpatient claims used for traditional agents and Medicare Part D dataset used for oral agents. Next lines of therapy were defined based on claims for new anticancer agents separated from the end of the prior line by some certain number of days. Sensitivity analyses were performed varying the number of days between therapies. Overall survival (OS) was defined as initiation of initial therapy to death, while OS2 was defined as time from initiation of second-line therapy to death. Time to next therapy 1 (TTNT1) was defined as time from start of first-line therapy to death or start of second-line therapy, whichever came first. Cox models and Kaplan-Meier (KM) survival curves were generated for time-to-event analyses. Analyses were stratified by categories of years of first- and second-line therapies corresponding to eras of key drug approvals (e.g., 2002-2005, 2006-2009, 2010-2013, >2013).
Results
In total, 4,832 patients were included, among whom 3,840 patients (79.5%) had claims for first-line regimens and 1,648 (34.1% of total and 42.9% of patients with first-line regimens) had second-line regimens. Of the 4,832 patients, the majority were male (67.4%), White (94.6%), and non-Hispanic (94.7%), with good performance status (74.8%) and a Charlson Comorbidity Index <2 (64.8%). The median age at diagnosis was 75 (IQR 70-81). Patients with first- and/or second-line regimens did not differ significantly from the entire population of 4,832 patients with MCL in terms of baseline characteristics.
In the first-line setting, use of bendamustine-rituximab (BR) increased from 2.3% of patients treated in 2006-2009 to 49.5% in 2014-2017. Similarly, use of single-agent BTK-inhibitors (BTKi) increased from nonexistent in patients treated in 2010-2013 to 13.8% in 2014-2017. In the second-line setting, use of single-agent BTKi similarly increased from 2.6% of those treated in 2010-2013 to 44.9% in 2014-2017.
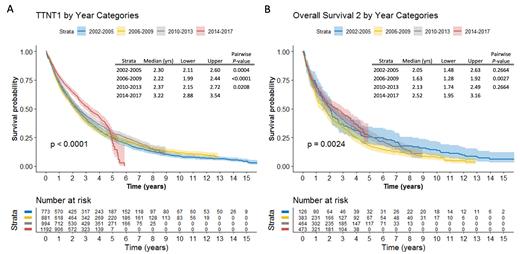
Median OS for the 3,840 patients with fist-line regimens was 4.4 years (95% CI, 4.1-4.5); median OS for those receiving first-line therapy in 2002-2005 was 3.5 years (3.1-3.9), compared to 3.7 (3.4-4.0), 4.5 (4.2-5.0), and not reached (4.8-NR) among those treated in 2006-2009, 2010-2013, and 2014-2017, respectively. Patients treated in 2014-2017 had significantly improved OS compared with those in 2002-2005 (log rank p<0.0001). Median TTNT1 overall was 2.5 years (2.4-2.6); median TTNT1 was 3.2 years (2.9-3.5) vs 2.3 years (2.1-2.6) for patients who received first-line therapy in 2014-2017 compared to 2002-2005 (log rank p<0.0001) ( Figure A). In addition, there was a significant difference across KM curves for OS2 stratified by categories of years of second-line therapy, with median OS2 of 2.5 (2.0-3.2) in patients treated in 2014-2017 compared to median OS2 of 2.1 (1.7-2.5) in 2010-2013 ( Figure B).
Discussion
In this analysis, we observed improvements survival of MCL patients over time, along with similar improvements in outcomes following first-line therapy, which may correlate with increased BR and BTKi use as first-line agents. Unexpectedly, improvements in survival following first-line therapy were considerably more modest. Potential explanations include lack of data from recent years during which we expect continued increased utilization of oral therapies vs unique impact of repeated therapies in the elderly. Overall, these data support the need for ongoing development of first- and subsequent-line therapies in MCL.
Disclosures
Wang:Innocare: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Novartis: Research Funding; Morphosys: Research Funding; Eli Lilly: Membership on an entity's Board of Directors or advisory committees, Research Funding; Incyte: Membership on an entity's Board of Directors or advisory committees, Research Funding; LOXO Oncology: Membership on an entity's Board of Directors or advisory committees, Research Funding; TG Therapeutics: Membership on an entity's Board of Directors or advisory committees; Kite: Honoraria, Membership on an entity's Board of Directors or advisory committees; Genmab: Research Funding; Genentech: Research Funding; Janssen: Membership on an entity's Board of Directors or advisory committees; Astra Zeneca: Membership on an entity's Board of Directors or advisory committees; BeiGene: Membership on an entity's Board of Directors or advisory committees; AbbVie: Consultancy. Maurer:Adaptive Biotechnologies: Membership on an entity's Board of Directors or advisory committees; AstraZeneca: Membership on an entity's Board of Directors or advisory committees; Roche/Genentech: Research Funding; BMS: Consultancy, Research Funding; GenMab: Membership on an entity's Board of Directors or advisory committees, Research Funding. Kahl:Abbvie: Consultancy, Honoraria; ADCT: Consultancy, Honoraria, Research Funding; BeiGene: Consultancy, Honoraria, Research Funding; BMS: Consultancy, Honoraria; Astra Zeneca: Consultancy, Honoraria, Research Funding; Janssen: Consultancy, Honoraria; Genmab: Consultancy, Honoraria; Genentech: Consultancy, Honoraria, Research Funding; Gilead: Consultancy, Honoraria; Lilly: Consultancy, Honoraria. Leonard:AbbVie, AstraZeneca, Astellas, Bayer, BeiGene, BMS, Calithera, Constellation, Eisai, Epizyme, GenMab, Grail, Incyte, Janssen, Karyopharm, Lilly, Merck, Mustang Bio, Pfizer, Roche/Genentech, Seagen, Second Genome, Sutro: Consultancy; National Cancer Institute, Leukemia and Lymphoma Society, Genentech, Epizyme, Janssen: Research Funding. Martin:AbbVie, AstraZeneca, Beigene, Epizyme, Genentech, Gilead, Janssen, Pepromene, Daiichi Sankyo: Consultancy.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal